Product Details
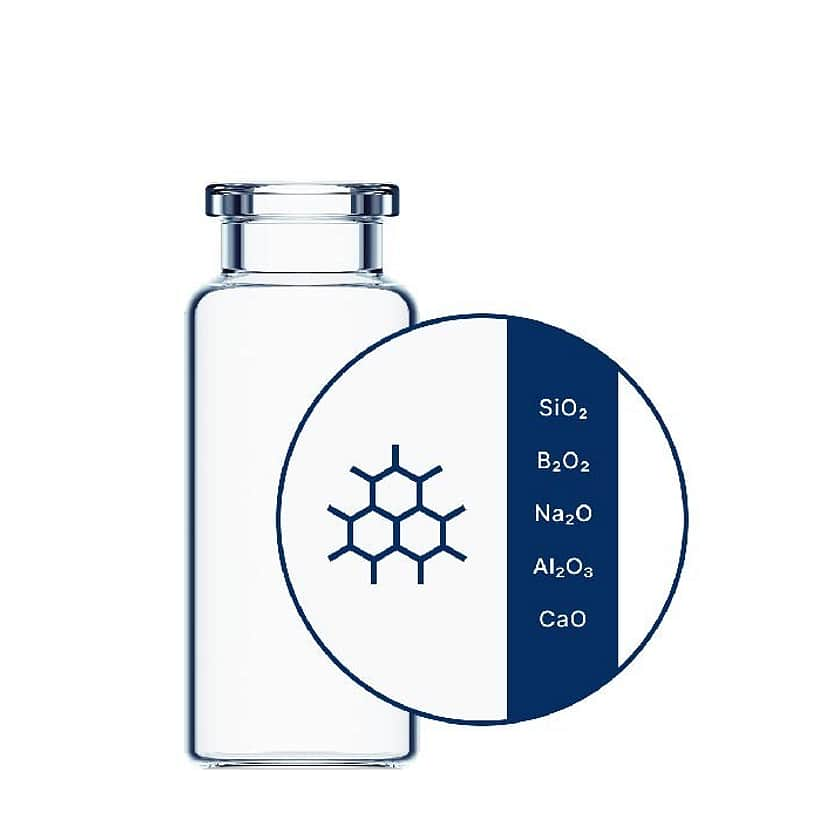
Ensure drug stability and avoid costly reformulations with ultra-pure vials by SCHOTT Pharma. EVERIC® pure features a homogenous inner surface area which protects against leachables, PH shifts and delamination, making it ideal for almost all drug formulations.
Product Applications
EVERIC® pure vials are designed to package challenging or expensive drug formulations where stability assurance is essential. Particularly suited to delamination-prone, low-fill biotech and biologics formulations, EVERIC® pure is also ideal for high-value pharmaceuticals and personalised medicines.